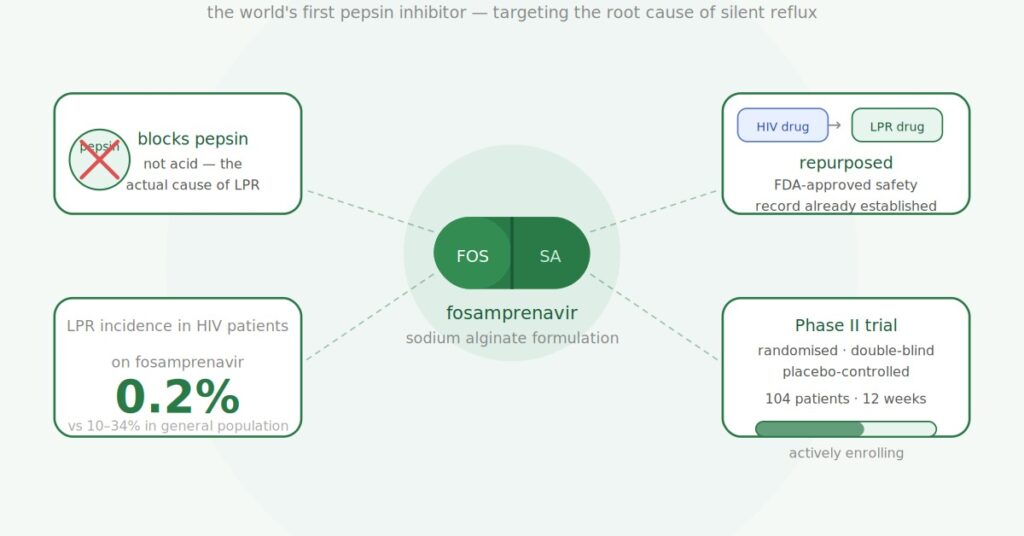
If you have LPR (silent reflux) and you’ve tried PPIs without much success — which is most people with LPR — there’s a genuinely exciting new treatment in development that works in a completely different way from anything currently available. It’s called fosamprenavir, and it’s the world’s first drug designed specifically to inhibit pepsin rather than acid.
This is significant. As I’ve explained throughout this site, pepsin — not acid — is the primary driver of LPR symptoms. Acid-suppressing medications like PPIs don’t touch pepsin. They reduce the acidity of reflux but the reflux still happens, and the pepsin in that reflux still reaches your throat and causes damage. Fosamprenavir targets that pepsin directly. It’s a fundamentally different approach and the early evidence behind it is compelling.
In this article I’ll explain what fosamprenavir is, where the idea came from, how it works, what the research shows so far, and where the treatment currently stands in development.
Key Takeaways:
- Fosamprenavir is an existing FDA-approved HIV drug being repurposed as the world’s first pepsin inhibitor for LPR
- It works by directly binding to and inactivating pepsin — the digestive enzyme responsible for most LPR throat damage
- The key epidemiological signal: among 2,062 HIV patients taking fosamprenavir, only 0.2% had documented LPR — versus 10 to 34% in the general population
- A mouse model study (Johnston et al., Laryngoscope 2023) showed both oral and inhaled fosamprenavir reversed pepsin-induced laryngeal damage
- Phase II clinical trials for LPR (and GERD) are currently underway, led by Dr. Nikki Johnston at the Medical College of Wisconsin through her company N-Zyme Biomedical
- An inhaled dry powder inhaler version is also in development — shown effective at 1/20th of the oral dose in preclinical studies
- This is not yet available as a treatment — it is in clinical trials
Why a New LPR Treatment Is Needed
Before getting into fosamprenavir specifically it’s worth understanding why this research matters so much — because it’s a direct response to a problem that anyone with LPR knows well.
PPIs are the standard treatment prescribed for LPR. The problem is they don’t work particularly well for it. Studies consistently show that around 86% of people with LPR don’t adequately respond to PPI therapy. The reason is simple: PPIs reduce the acidity of reflux but they don’t stop reflux from happening. And for LPR, it’s not just the acid that matters — it’s the pepsin travelling up with it.
Pepsin is a digestive enzyme produced in the stomach. When it refluxes into the throat it causes inflammation and tissue damage — and it can do this even when the reflux isn’t particularly acidic. Pepsin can also lie dormant in throat cells for 24 to 48 hours, then be reactivated by anything acidic that comes into contact with it. This is the cycle that keeps LPR going even when patients are on maximum acid suppression.
Despite this being well established in the research for over a decade, there has been no drug specifically designed to target pepsin. Until now. You can read more about why pepsin matters so much in my complete guide to LPR.
What Is Fosamprenavir?
Fosamprenavir (brand name Lexiva in the US, Telzir in Europe) is an existing FDA-approved protease inhibitor used to treat HIV. It’s been approved and used safely in humans for decades as part of HIV treatment regimens, which is one of the things that makes it an attractive candidate for repurposing — the safety profile is already well established.
The discovery that it might work for LPR came from Dr. Nikki Johnston, a researcher at the Medical College of Wisconsin who has spent over a decade studying pepsin’s role in reflux disease. Her lab identified that certain HIV protease inhibitors — drugs designed to block a specific enzyme in the HIV virus — also happen to bind to and inhibit pepsin. This makes sense mechanistically: fosamprenavir works against HIV by blocking a viral protease enzyme, and pepsin is also a protease enzyme with a structurally similar active site. The drug essentially doesn’t distinguish between the two.
The epidemiological signal that followed this discovery was striking. When Dr. Johnston’s team looked at the records of 2,062 adult HIV patients who had been prescribed HIV protease inhibitors at Froedtert Memorial Lutheran Hospital, only 0.2% had documented LPR — compared to an incidence of 10 to 34% in the general population. That’s a massive disparity and it strongly suggests the protease inhibitors were inadvertently protecting these patients from LPR.
How Fosamprenavir Works for LPR
Fosamprenavir works as a pepsin inhibitor — it directly binds to pepsin and blocks its enzymatic activity. Here’s the mechanism in plain terms:
1. Fosamprenavir is converted to amprenavir in the body — fosamprenavir is a prodrug, meaning it’s inactive until the body metabolises it. Once absorbed it converts to amprenavir, which is the active form that binds to and inhibits pepsin.
2. Amprenavir binds directly to pepsin — it attaches to pepsin’s active site, the part of the enzyme that causes damage, and blocks it from functioning. This prevents pepsin from triggering the inflammation and tissue damage in the larynx and throat that causes LPR symptoms.
3. The damage cycle is broken — rather than reducing acid production and hoping pepsin does less damage as a result (the PPI approach), fosamprenavir directly neutralises the pepsin itself. This addresses the actual mechanism of injury in LPR rather than an indirect contributing factor.
Laboratory studies have confirmed amprenavir inhibits pepsin at low micromolar concentrations — concentrations that are achievable in the body at the manufacturer’s recommended dose for HIV treatment. A 2023 in vitro study (Samuels et al.) showed amprenavir protected laryngeal epithelial cells from pepsin-induced barrier disruption and prevented the molecular changes associated with cancer progression — including E-cadherin cleavage and matrix metalloproteinase induction.
A 2025 study by Ergun et al. in the International Journal of Molecular Sciences further confirmed that amprenavir mitigates pepsin-induced transcriptomic changes in both normal and precancerous oesophageal cells — meaning it may protect against the cancer-associated cellular changes that persistent pepsin exposure can trigger.
What the Animal Model Study Showed
The most important preclinical study to date is Johnston et al. (Laryngoscope 2023), which tested fosamprenavir in a mouse model specifically designed to replicate the laryngeal damage seen in LPR patients.
Mice were exposed to pepsin instilled directly into the larynx — replicating what happens during reflux episodes in LPR. The untreated mice developed the reactive epithelial changes, increased inflammatory cells and cell apoptosis (cell death) characteristic of LPR tissue damage. The mice treated with fosamprenavir — both oral and inhaled versions — showed normal laryngeal histology despite the pepsin exposure. The damage was effectively prevented.
Two delivery methods were tested:
- Oral fosamprenavir at the human-equivalent dose completely prevented the laryngeal damage
- Inhaled fosamprenavir delivered directly to the larynx was effective at 1/20th of the oral dose — a significant finding that points toward a much lower-dose inhaler formulation being viable
This was the study that provided proof of concept in vivo — confirming that the drug-target interaction observed in the lab actually translates to protection of laryngeal tissue in a living model.
The Two Formulations Being Developed
N-Zyme Biomedical — the company founded by Dr. Johnston and business partner Franco Vigile specifically to develop this treatment — is working on two distinct formulations of fosamprenavir for reflux:
1. Oral fosamprenavir-sodium alginate (FOS-SA)
This is a sustained-release oral formulation that combines fosamprenavir with sodium alginate — yes, the same ingredient that makes Gaviscon Advance so effective. The sodium alginate is included to increase mucoadhesion, meaning the drug sticks to the oesophageal lining longer and provides extended local delivery rather than being absorbed rapidly into the bloodstream. This is the formulation being tested in the current Phase II clinical trial for LPR, and a separate Phase II trial for GERD is estimated to start in September 2026.
2. Inhaled dry powder inhaler (DPI)
The inhaled version delivers fosamprenavir directly to the larynx and throat — the site of LPR damage — at much lower doses than oral administration. A 2024 study (Lesnick et al.) assessed the safety of inhaled fosamprenavir in a rodent model over four weeks and found a good safety profile across all organs examined. Computational fluid dynamics modelling identified an optimal particle size of 9 to 12 micrometres for deposition in the human laryngopharynx. A formal Good Laboratory Practice (GLP) toxicology assessment is underway to support a Phase I clinical trial of the inhaler.
The inhaler formulation is particularly exciting for LPR patients because it would deliver the drug exactly where the damage is occurring, at far lower systemic doses, which should mean an even better side effect profile than the oral version.
The Clinical Trial
The current Phase II trial (registered at ClinicalTrials.gov as NCT04383262) is a 12-week randomised, double-blind, placebo-controlled trial testing oral fosamprenavir-sodium alginate in patients with moderate to severe LPR who have already failed PPI treatment.
Key details:
- 104 participants total — 52 receiving fosamprenavir-sodium alginate, 52 receiving placebo
- Participants must have a confirmed LPR diagnosis by pH impedance monitoring, an RSI score above 13, and documented failure of at least 3 months of twice-daily PPI therapy
- Primary outcomes are measured using the Reflux Symptom Index (RSI), Reflux Finding Score (RFS) and Voice Handicap Index — the standard clinical outcome measures for LPR
- Led by Dr. Nikki Johnston at the Medical College of Wisconsin
- Funded by venture capital investment in N-Zyme Biomedical and by the Medical College of Wisconsin
The trial is targeting exactly the population most underserved by current treatments — people with confirmed LPR who have already tried PPIs and not improved. If positive results emerge from this trial it would be the first clinical evidence of an effective medical therapy specifically for LPR.
When Could Fosamprenavir Be Available as an LPR Treatment?
This is the important question to be realistic about. Fosamprenavir for LPR is currently in Phase II clinical trials — which tests efficacy and safety in a controlled patient population. After Phase II, assuming positive results, Phase III trials (larger scale) would be required before any regulatory approval process could begin.
Realistically this means fosamprenavir as an approved LPR treatment is likely several years away at minimum — probably not before 2028 to 2030 at the earliest under a normal drug development timeline, and potentially longer. Drug development timelines are notoriously unpredictable.
That said, the repurposing approach here is genuinely advantageous. Because fosamprenavir is already FDA-approved with a well-established safety record, the regulatory pathway is considerably faster than for a brand new compound. The Phase II trial doesn’t need to establish basic safety from scratch — that’s already done. This could meaningfully compress the timeline compared to a novel drug.
It’s also worth noting that fosamprenavir is an existing drug available now — some people have asked whether it’s possible to obtain it for LPR off-label before trial results are available. I’d advise against this without specialist guidance. The dose and formulation being tested for LPR is different from the HIV formulation, the drug has interactions and contraindications that need to be properly assessed, and there is no clinical evidence yet on efficacy specifically for LPR in humans. Wait for the trial results.
Why This Matters for LPR Patients
For anyone who has been through the frustration of trying PPIs that don’t work, being told your symptoms might be anxiety or stress, or just not being able to get a proper diagnosis or effective treatment — this research matters because it represents the first genuine attempt to develop a medication that targets what actually causes LPR.
The entire treatment paradigm for LPR for the past two decades has been acid suppression — a blunt instrument that addresses the wrong target. Fosamprenavir would be the first drug to directly address pepsin, which is the right target. If the clinical trials confirm what the preclinical data suggests, this could fundamentally change how LPR is treated.
In the meantime — while the trials run and the treatment develops — the best approach remains the diet, Gaviscon Advance and lifestyle changes that address LPR through the same principle of controlling pepsin exposure. You can read about the complete treatment approach in my silent reflux treatment guide and the structured diet plan in the Wipeout Diet Plan. I’ll update this article as the trial progresses and results become available.
Frequently Asked Questions
What is fosamprenavir for LPR?
Fosamprenavir is an existing FDA-approved HIV drug being repurposed as the world’s first pepsin inhibitor for the treatment of LPR (silent reflux). It works by directly binding to and inhibiting pepsin — the digestive enzyme responsible for most of the throat and laryngeal damage in LPR. It’s currently in Phase II clinical trials led by Dr. Nikki Johnston at the Medical College of Wisconsin through N-Zyme Biomedical.
How does fosamprenavir work differently from PPIs?
PPIs reduce stomach acid production but don’t prevent reflux from occurring, and don’t affect pepsin at all. Fosamprenavir directly inhibits pepsin — blocking its ability to cause inflammation and damage in the throat regardless of the acidity of the reflux. This is the fundamental difference: PPIs target acid, fosamprenavir targets pepsin, which is the actual cause of LPR tissue damage.
Is fosamprenavir available now for LPR?
No — it is currently in Phase II clinical trials only and is not approved or widely available as an LPR treatment. While fosamprenavir exists as an approved HIV drug, the formulation being developed for LPR (fosamprenavir-sodium alginate) is a new formulation and the human evidence for LPR efficacy is not yet established. Clinical trial results are expected within the next few years.
What is N-Zyme Biomedical?
N-Zyme Biomedical is the biotech company founded by Dr. Nikki Johnston (Chief Scientific Officer) and Franco Vigile (CEO) to develop fosamprenavir as the first pepsin inhibitor for LPR and GERD. The company is based in Milwaukee, Wisconsin and is funded by venture capital investment. They are also developing an inhaled dry powder inhaler version of the drug for direct laryngopharyngeal delivery.
Why is it being developed from an HIV drug?
Fosamprenavir was identified as a pepsin inhibitor because HIV protease inhibitors — the class of drugs fosamprenavir belongs to — work by blocking a viral protease enzyme. Pepsin is also a protease enzyme with a structurally similar active site, so these drugs inadvertently bind to pepsin as well. The key epidemiological evidence: among 2,062 HIV patients taking protease inhibitors, only 0.2% had documented LPR versus 10 to 34% in the general population — suggesting the drug was inadvertently protecting them from LPR.
What is the inhaled version of fosamprenavir?
N-Zyme Biomedical is also developing a dry powder inhaler (DPI) version of fosamprenavir that delivers the drug directly to the larynx and throat — the site of LPR damage — rather than systemically via the bloodstream. In the mouse model study, inhaled fosamprenavir was effective at just 1/20th of the oral dose. A GLP toxicology assessment is underway and a Phase I clinical trial of the inhaler is planned.
Should I try to get fosamprenavir now for my LPR?
No — I’d advise against attempting to obtain fosamprenavir off-label for LPR at this stage. The specific formulation being tested for LPR is different from the standard HIV formulation, the drug has significant interactions and contraindications, and there are no human clinical trial results yet confirming efficacy or appropriate dosing for LPR. The best current approach remains the low acid diet and Gaviscon Advance while the trials proceed.
Conclusion
Fosamprenavir for LPR is the most scientifically compelling new treatment in development for silent reflux — and that’s not an overstatement. It’s the first drug to actually target the right thing. The preclinical evidence is strong, the epidemiological signal is striking, and the Phase II trial is actively running in exactly the patient population that needs it most — people who have failed PPIs and have no other effective medical option.
It’s not available yet and it’s worth being patient rather than trying to access it prematurely. But if the trial results confirm what the early data suggests, this could genuinely change the treatment landscape for LPR in a way that hasn’t happened in decades.
I’ll be keeping a close eye on the trial progress and will update this article as results emerge. In the meantime, for the most effective current treatment approach check my silent reflux treatment guide, the LPR diet guide, and the Wipeout Diet Plan. And if you want personalised guidance on your specific situation consider a private consultation.
Related articles:
- The Complete Guide to LPR — Causes, Symptoms and Treatment
- Silent Reflux Treatment — What Actually Works for LPR
- GERD vs LPR — What’s the Difference?
- Gaviscon Advance for LPR — Why the UK Version Is the Only One That Works
- LPR Diet — What to Eat, What to Avoid and What to Drink
- LPR Symptoms — The Complete Guide
- Peptest Review — Is It Worth Doing for LPR?
- How I Cured My Silent Reflux
References
- Johnston N et al. (2023) “Oral and Inhaled Fosamprenavir Reverses Pepsin-Induced Damage in a Laryngopharyngeal Reflux Mouse Model.” The Laryngoscope, 133 Suppl 1:S1-S11. PMC
- Lesnick A et al. (2024) “Inhaled fosamprenavir for laryngopharyngeal reflux: Toxicology and fluid dynamics modeling.” Laryngoscope Investigative Otolaryngology, 9(1):e1219. PMC
- Samuels TL et al. (2023) “Amprenavir inhibits pepsin-mediated laryngeal epithelial disruption and E-cadherin cleavage in vitro.” Laryngoscope Investigative Otolaryngology, 8(4):953-962. PubMed
- Ergun P, Samuels TL, Johnston N et al. (2025) “Amprenavir Mitigates Pepsin-Induced Transcriptomic Changes in Normal and Precancerous Esophageal Cells.” International Journal of Molecular Sciences, 26(13):6182. PubMed
- ClinicalTrials.gov. “Fosamprenavir for Laryngopharyngeal Upper Airway Tissue Treatment and Enzyme Reduction.” NCT04383262. ClinicalTrials.gov
- Medical College of Wisconsin. “New Approach to Reflux Treatment Could Revolutionize Care for LPR and GERD.” MCW
- N-Zyme Biomedical. Research overview. nzymebio.com